The experimental measurement of the heat of reaction or enthalpy change is known as colarimetry.
The name evidently finds its origin in the unit of heat- the -caloric. The heat given out or absorbed in a chemical reaction is measured in a suitable apparatus called a calorimeter. These calorimeter vary considerably in their construction and designs. The are adapted to suit the requirements of a particular reaction under study. For instance, to measure the heats of reactions involving (i). Solutions only, (ii). Gases, (iii). Very reactive chemical etc., different types of calorimeters are employed. We discuss below two of the common type of calorimeters.
Water Calorimeter
This is convenient apparatus for finding the heat changes accompanying chemical reactions taking place in solutions.
The apparatus consists essentially of a water-bath with thermally insulated walls. A reaction chamber consisting of two limbs is suspended in the water-bath. Through the lid of the water-bath pass (a). thermometer that records the temputure variation and (b). A stirrer that stirs water in the water-bath.
A known quantity of water (say w gms) is taken in the water-bath and its temputure is noted. The reacting substances are filled in the two limbs. The reacting chamber is now turned upside down (position ll) to allow the solution to mix. They react and the heat roduced during the reaction is taken up by water, raising its temputure. If the rise in temperature (Final reading - Initial reading) is °C, the heat absorbed by water 'Q' is given by
Q=W×1×t caloies
But heat produced in the reaction is equal to the absorbed by water. hence of the reaction can be calculated.
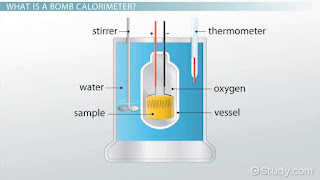
Bomb Calorimeter
This apparatus was devised by Berthelot (1881) to measure the heat of combustion of organic compounds. A modified from of the apparatus consists of a sealed combustion chamber, called a bomb, containing a weighed quantity of the substances in a dish along with oxygen under about 2026500.00 pascal pressure . The bomb is lowered in water contained in an insulated copper vessel. This vessel is provided with a stirrer and a thermometer reading up to 1/100th of a degree. It is also surrounded by an outer jacket to ensure complete insulation from the atmosphere. The temperature of water is noted before the substances is ignited by an electric current. After combustion, the rise in temperature of the system is noted on the thermometer and heat of combustion can be calculated from the heat gained by water and the calorimeter.